china umbilical cord plastic clamp
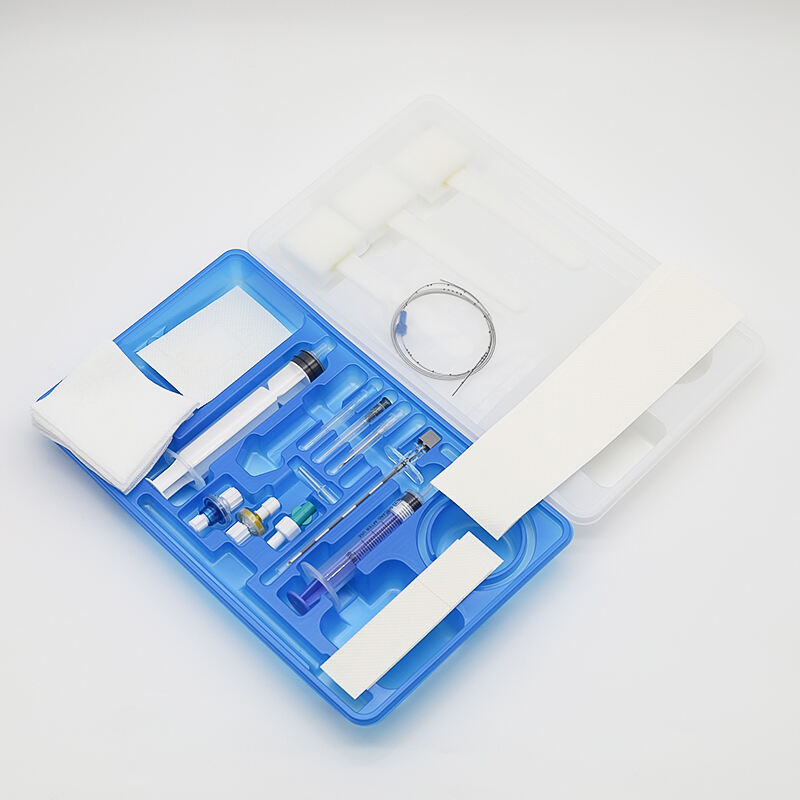
The china umbilical cord plastic clamp represents a critical medical device designed to ensure safe and effective cord clamping procedures immediately following birth. This specialized surgical instrument serves as the primary method for securing the umbilical cord, preventing blood loss and establishing proper circulation for newborns. The china umbilical cord plastic clamp functions by creating a secure, airtight seal around the cord tissue, effectively stopping blood flow between mother and infant during the crucial post-delivery period. Healthcare professionals rely on this essential tool to perform one of the most fundamental procedures in obstetric care. The technological features of the china umbilical cord plastic clamp include precision-engineered plastic construction that provides optimal grip strength while maintaining gentle pressure on delicate cord tissue. The device incorporates ergonomic design elements that facilitate easy handling during high-stress delivery situations. Advanced manufacturing processes ensure consistent quality and reliability across all units produced. The clamp mechanism utilizes a spring-loaded system that automatically adjusts to varying cord diameters, accommodating different patient anatomies without compromising effectiveness. Surface texturing on the gripping areas enhances security and prevents slippage during application. The china umbilical cord plastic clamp finds extensive applications across multiple healthcare settings, including hospitals, birthing centers, ambulatory surgical facilities, and emergency medical services. Obstetricians, midwives, nurses, and emergency responders utilize these devices during routine deliveries, emergency births, and cesarean procedures. The versatility of the china umbilical cord plastic clamp makes it suitable for various clinical scenarios, from planned hospital births to unexpected emergency situations. Its compact design enables easy storage and transport, making it an essential component of mobile medical kits and delivery room equipment. The device plays a crucial role in infection control protocols, as its single-use design eliminates cross-contamination risks between patients.